Beer's Law Factors . factors used to derive the beer’s law. This fractional decrease in power is proportional to the sample’s thickness and to the. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. beer’s law is a limiting law that is valid only for low concentrations of analyte. There are two contributions to this fundamental. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. The premise is that a light. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can.
from studylib.net
There are two contributions to this fundamental. The premise is that a light. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. factors used to derive the beer’s law. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. This fractional decrease in power is proportional to the sample’s thickness and to the. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer’s law is a limiting law that is valid only for low concentrations of analyte.
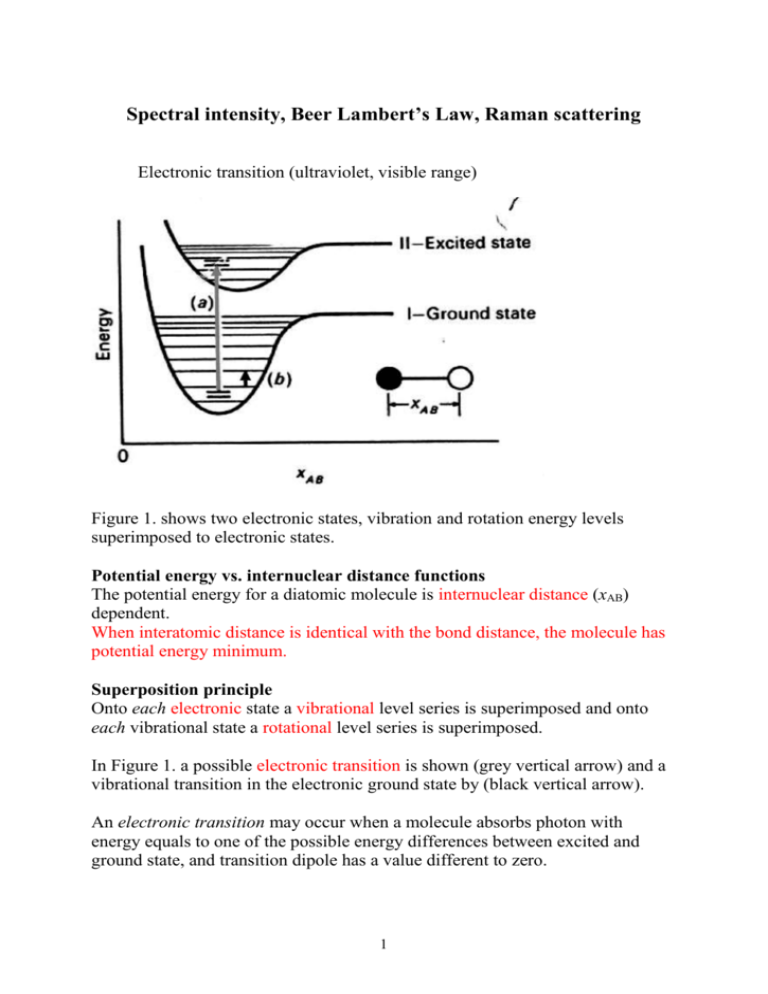
Beer Lambert`s law, Raman spectroscopy
Beer's Law Factors This fractional decrease in power is proportional to the sample’s thickness and to the. The premise is that a light. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. This fractional decrease in power is proportional to the sample’s thickness and to the. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. beer’s law is a limiting law that is valid only for low concentrations of analyte. factors used to derive the beer’s law. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. There are two contributions to this fundamental.
From egpat.com
Deviations from BeerLambert law Beer's Law Factors beer's law states that a chemical solution's concentration is directly proportional to its light absorption. This fractional decrease in power is proportional to the sample’s thickness and to the. There are two contributions to this fundamental. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. since the concentration, path length. Beer's Law Factors.
From www.slideserve.com
PPT Exp 14B Determining an Equilibrium Constant PowerPoint Beer's Law Factors This fractional decrease in power is proportional to the sample’s thickness and to the. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. There are two contributions to this fundamental. The premise is that a light. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we. Beer's Law Factors.
From studylib.net
Beer`s Law Beer's Law Factors This fractional decrease in power is proportional to the sample’s thickness and to the. There are two contributions to this fundamental. beer’s law is a limiting law that is valid only for low concentrations of analyte. factors used to derive the beer’s law. beer's law states that a chemical solution's concentration is directly proportional to its light. Beer's Law Factors.
From thechemistrynotes.com
BeerLambert Law Statement, Derivation, Applications, Limitations Beer's Law Factors There are two contributions to this fundamental. This fractional decrease in power is proportional to the sample’s thickness and to the. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. The premise is that a light. factors used to derive the beer’s law. beer's law states that a chemical. Beer's Law Factors.
From www.youtube.com
Beer's Law Overview YouTube Beer's Law Factors beer’s law is a limiting law that is valid only for low concentrations of analyte. There are two contributions to this fundamental. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. factors used to derive the beer’s law. The premise is that a light. since the concentration, path length and. Beer's Law Factors.
From www.scienceabc.com
Beers Law Definition, History, Equation, Formula And Example Beer's Law Factors beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. The premise is that a light. This fractional decrease. Beer's Law Factors.
From www.studocu.com
Chem 181 4 Beer's Law Beer Lab Pre lab Determining the Beer's Law Factors since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. factors used to derive the beer’s law. The premise is that a light. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. There are two contributions to this fundamental. beer’s law is. Beer's Law Factors.
From www.slideserve.com
PPT Atmospheric Transmission PowerPoint Presentation, free download Beer's Law Factors beer’s law is a limiting law that is valid only for low concentrations of analyte. There are two contributions to this fundamental. The premise is that a light. This fractional decrease in power is proportional to the sample’s thickness and to the. factors used to derive the beer’s law. since the concentration, path length and molar absorptivity. Beer's Law Factors.
From www.youtube.com
BeerLambert law in easy way YouTube Beer's Law Factors The premise is that a light. This fractional decrease in power is proportional to the sample’s thickness and to the. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. factors used to derive the beer’s law. beer’s law is a limiting law that is valid only for low concentrations of. Beer's Law Factors.
From www.chegg.com
Solved a. ( 3 pts.) Use Beer's Law and the directions above Beer's Law Factors beer's law states that a chemical solution's concentration is directly proportional to its light absorption. The premise is that a light. This fractional decrease in power is proportional to the sample’s thickness and to the. beer’s law is a limiting law that is valid only for low concentrations of analyte. since the concentration, path length and molar. Beer's Law Factors.
From www.thoughtco.com
Beer's Law Definition and Equation Beer's Law Factors The premise is that a light. There are two contributions to this fundamental. This fractional decrease in power is proportional to the sample’s thickness and to the. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer's law states that a chemical solution's concentration is directly proportional to its light. Beer's Law Factors.
From ceduudiv.blob.core.windows.net
Beer's Law Solve For C at Wyatt Edgell blog Beer's Law Factors The premise is that a light. There are two contributions to this fundamental. beer’s law is a limiting law that is valid only for low concentrations of analyte. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. factors used to derive the beer’s law. since the concentration, path length and. Beer's Law Factors.
From www.slideserve.com
PPT chapter 2 PowerPoint Presentation, free download ID540228 Beer's Law Factors There are two contributions to this fundamental. beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. beer’s law is a limiting law that is valid only for low concentrations of analyte. The premise is that a light. This fractional decrease in power is proportional to the sample’s thickness and to the.. Beer's Law Factors.
From www.slideserve.com
PPT Beer’s Law & Colorimetry PowerPoint Presentation, free download Beer's Law Factors The premise is that a light. This fractional decrease in power is proportional to the sample’s thickness and to the. factors used to derive the beer’s law. There are two contributions to this fundamental. beer’s law is a limiting law that is valid only for low concentrations of analyte. beer’s law, in spectroscopy, a relation concerning the. Beer's Law Factors.
From www.vernier.com
Determining the Concentration of a Solution Beer's Law > Experiment 17 Beer's Law Factors since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer’s law is a limiting law that is valid only for low concentrations of analyte. This fractional decrease in power is proportional to the sample’s thickness and to the. factors used to derive the beer’s law. The premise is that. Beer's Law Factors.
From www.slideserve.com
PPT Beers Law for a Single Component Sample PowerPoint Presentation Beer's Law Factors beer’s law, in spectroscopy, a relation concerning the absorption of radiant energy by an absorbing medium. factors used to derive the beer’s law. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer's law states that a chemical solution's concentration is directly proportional to its light absorption. The. Beer's Law Factors.
From calculatorghw.blogspot.com
Beer Lambert Law Calculator CALCULATOR GHW Beer's Law Factors The premise is that a light. since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. beer’s law is a limiting law that is valid only for low concentrations of analyte. factors used to derive the beer’s law. beer's law states that a chemical solution's concentration is directly proportional. Beer's Law Factors.
From exolwzeip.blob.core.windows.net
Beer's Law Equation Chemistry at Taylor Jones blog Beer's Law Factors since the concentration, path length and molar absorptivity are all directly proportional to the absorbance, we can. factors used to derive the beer’s law. beer’s law is a limiting law that is valid only for low concentrations of analyte. This fractional decrease in power is proportional to the sample’s thickness and to the. beer’s law, in. Beer's Law Factors.